A quality by design approach in oral extended release drug delivery systems: where we are and where we are going?

Oral extended release (ER) delivery systems have quickly gained increasing importance because of their ability to maintain drug levels in the blood more consistently, reducing side effects and improving patient compliance. The complexity of ER formulation leads to additional development challenges in the fulfilment of quality-related regulatory requirements. Despite their challenging properties, the potential of ER system formulation and process development can be better exploited by applying quality by design (QbD) approaches and advanced modeling techniques such as machine learning (ML).
Area covered
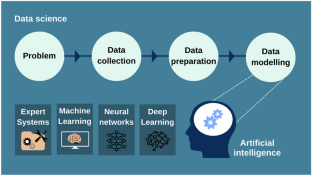
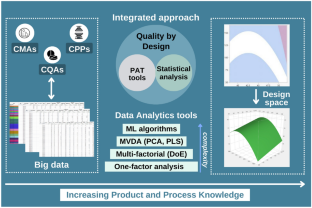
This review provides a comprehensive overview of QbD concepts applied to oral ER delivery systems, clarifying the impact of raw materials and process variables on critical quality attributes (CQAs). Moreover, data science coupled with ML algorithms is also elucidated in this article as a potential tool for predicting and optimizing ER formulation design and manufacturing processes.
Expert opinion
QbD, as a scientific and risk-based approach, provides a comprehensive understanding of oral ER drug delivery systems improving product quality and reducing postapproval changes. Enabling QbD with ML-driven pharmaceutical development can provide an opportunity to move toward risk mitigation for efficient ER tablet formulation and process development. However, there are some barriers to overcome in the way of adopting QbD concepts. The key issues are the lack of understanding and the gap between industries and regulatory authorities concerning the scientific principles and terms beyond QbD, which can create an obstacle during the approval process. Furthermore, it is generally believed that the resources and time invested in applying QbD tools are not cost-effective during constant and continuous improvement. Today, it is time to realize that a multidisciplinary understanding of the formulation and manufacturing process is as important as achieving the final result.
This is a preview of subscription content, log in via an institution to check access.
Access this article
Subscribe and save
Springer+ Basic
€32.70 /Month
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
Buy Now
Price includes VAT (France)
Instant access to the full article PDF.
Rent this article via DeepDyve
Similar content being viewed by others
Application of Absorption Modeling in Rational Design of Drug Product Under Quality-by-Design Paradigm
Article 22 May 2015
Product Development, Manufacturing, and Packaging of Solid Dosage Forms Under QbD and PAT Paradigm: DOE Case Studies for Industrial Applications
Article 16 September 2019
Scientific and Regulatory Considerations in Solid Oral Modified Release Drug Product Development
Article 20 September 2016
Explore related subjects
References
- Abu Fara D, Dadou SM, Rashid I, Al-Obeidi R, Antonijevic MD et al (2019) A direct compression matrix made from xanthan gum and low molecular weight chitosan designed to improve compressibility in controlled release tablets. Pharmaceutics 11:603 ArticleCASPubMedPubMed CentralGoogle Scholar
- Aguilar-De-Leyva A, Campinez MD, Casas M, Caraballo I (2017) Design space and critical points in solid dosage forms. J Drug Deliv Sci Technol 42:134–143 ArticleCASGoogle Scholar
- Akhtar MF, Ashraf H, Uzair M, Ahmad S, Rasul A et al (2022) Development of leachable enalapril tablets by controlled porosity osmotic pump technique; a unique approach to enhance its sustained release effect. J Coat Technol Res 19:497–507 ArticleCASGoogle Scholar
- Akseli I, Xie JJ, Schultz L, Ladyzhynsky N, Bramante T et al (2017) A practical framework toward prediction of breaking force and disintegration of tablet formulations using machine learning tools. J Pharm Sci 106:234–247 ArticleCASPubMedGoogle Scholar
- Aktas E, Eroglu H, Kockan U, Oner L (2013) Systematic development of pH-independent controlled release tablets of carvedilol using central composite design and artificial neural networks. Drug Dev Ind Pharm 39:1207–1216 ArticleCASPubMedGoogle Scholar
- Al-Zoubi N, Kachrimanis K, Younis K, Malamataris S (2011) Optimization of extended-release hydrophilic matrix tablets by support vector regression. Drug Dev Ind Pharm 37:80–87 ArticleCASPubMedGoogle Scholar
- Al-Zoubi N, Alkhatib HS, Alobaidi G, Abdel-Rahim S, Obeidat W et al (2015) Optimization of pH-independent chronotherapeutic release of verapamil HCl from three-layer matrix tablets. Int J Pharm 494:296–303 ArticleCASPubMedGoogle Scholar
- Arden S, Fisher AC, Tyner K, Yu LCX, Lee SL et al (2021) Industry 4.0 for pharmaceutical manufacturing: preparing for the smart factories of the future. Int J Pharm 602:120554 ArticleCASPubMedGoogle Scholar
- Ashenden SK, Bartosik A, Agapow P-M, Semenova E (2021) Introduction to artificial intelligence and machine learning. In: Ashenden S (ed) The era of artificial intelligence, machine learning, and data science in the pharmaceutical industry, 1st edn. Academic Press, United Kingdom, pp 15–26 ChapterGoogle Scholar
- Banner M, Alosert H, Spencer C, Cheeks M, Farid SS et al (2021) A decade in review: use of data analytics within the biopharmaceutical sector. Curr Opin Chem Eng 34:100758 ArticleGoogle Scholar
- Bannigan P, Aldeghi M, Bao ZQ, Hase F, Aspuru-Guzik A et al (2021) Machine learning directed drug formulation development. Adv Drug Deliv Rev 175:113806 ArticleCASPubMedGoogle Scholar
- Barenji RV, Akdag Y, Yet B, Oner L (2019) Cyber-physical-based PAT (CPbPAT) framework for Pharma 4.0. Int J Pharm 567:118445 ArticleCASPubMedGoogle Scholar
- Barmpalexis P, Kanaze FI, Kachrimanis K, Georgarakis E (2010) Artificial neural networks in the optimization of a nimodipine controlled release tablet formulation. Eur J Pharm Biopharm 74:316–323 ArticleCASPubMedGoogle Scholar
- Barmpalexis P, Kachrimanis K, Malamataris S (2018) Statistical moments in modelling of swelling, erosion and drug release of hydrophilic matrix-tablets. Int J Pharm 540:1–10 ArticleCASPubMedGoogle Scholar
- Benedetti A, Khoo J, Sharma S, Facco P, Barolo M et al (2019) Data analytics on raw material properties to accelerate pharmaceutical drug development. Int J Pharm 563:122–134 ArticleCASPubMedGoogle Scholar
- Bermejo M, Sanchez-Dengra B, Gonzalez-Alvarez M, Gonzalez-Alvarez I (2020) Oral controlled release dosage forms: dissolution versus diffusion. Expert Opin Drug Deliv 17:791–803 ArticleCASPubMedGoogle Scholar
- Bruschi ML (2015) Strategies to modify the drug release from pharmaceutical systems. Woodhead Publishing, United Kingdom Google Scholar
- Caccavo D, Cascone S, Lamberti G, Barba AA (2014) Modeling the drug release from hydrogel-based matrices. Mol Pharm 12:474–483 ArticlePubMedGoogle Scholar
- Chakraborty S, Khandai M, Sharma A, Patra CN, Patro VJ et al (2009) Effects of drug solubility on the release kinetics of water soluble and insoluble drugs from HPMC based matrix formulations. Acta Pharm 59:313–323 ArticleCASPubMedGoogle Scholar
- Chappidi SR, Bhargav E, Marikunte V, Chinthaginjala H, Vijaya Jyothi M et al (2019) A cost effective (QbD) approach in the development and optimization of rosiglitazone maleate mucoadhesive extended release tablets—in vitro and ex vivo. Adv Pharm Bull 9:281–288 ArticlePubMedPubMed CentralGoogle Scholar
- Chudiwal VS, Shahi S, Chudiwal S (2018) Development of sustained release gastro-retentive tablet formulation of nicardipine hydrochloride using quality by design (QbD) approach. Drug Dev Ind Pharm 44:787–799 ArticleCASPubMedGoogle Scholar
- Colombo P, Bettini R, Santi P, Peppas NA (2000) Swellable matrices for controlled drug delivery: gel-layer behaviour, mechanisms and optimal performance. Pharm Sci Technol Today 3:198–204 ArticleCASPubMedGoogle Scholar
- Costa P, Manuel J, Lobo S (2001) Modeling and comparison of dissolution profiles. Eur J Pharm Sci 13:123–133 ArticleCASPubMedGoogle Scholar
- Crowley MM, Schroeder B, Fredersdorf A, Obara S, Talarico M et al (2004) Physicochemical properties and mechanism of drug release from ethyl cellulose matrix tablets prepared by direct compression and hot-melt extrusion. Int J Pharm 269:509–522 ArticleCASPubMedGoogle Scholar
- Dave VS, Saoji SD, Raut NA, Haware RV (2015) Excipient variability and its impact on dosage form functionality. J Pharm Sci 104:906–915 ArticleCASPubMedGoogle Scholar
- Davis B, Schlindwein WS (2018) Introduction to quality by design (QbD). In: Schlindwein WS, Gibson M (eds) Pharmaceutical quality by design: a practical approach, 1st edn. John Wiley & Sons, United States, pp 1–9 Google Scholar
- Demchenko Y, Grosso P, de Laat C, Membrey P (2013) Addressing big data issues in scientific data infrastructure. International Conference on Collaboration Technologies and Systems (CTS), San Diego BookGoogle Scholar
- Desai N, Purohit R (2017) Design and development of clopidogrel bisulfate gastroretentive osmotic formulation using quality by design tools. AAPS PharmSciTech 18:2626–2638 ArticleCASPubMedGoogle Scholar
- Diab S, Bano G, Dhenge RM, Taylor J (2021) Impact of process parameters and formulation properties on dissolution performance of an extended release tablet: a multivariate analysis. J Pharm Innov 17:892–910 ArticleGoogle Scholar
- Ding H (2016) Modified-release drug products and drug devices. In: Shargel L, Yu AB (eds) Applied biopharmaceutics and pharmacokinetics, 7th edn. McGraw-Hill Education, New York Google Scholar
- Djuris J, Cirin-Varadjan S, Aleksic I, Djuris M, Cvijic S et al (2021) Application of machine-learning algorithms for better understanding of tableting properties of lactose co-processed with lipid excipients. Pharmaceutics 13:663 ArticleCASPubMedPubMed CentralGoogle Scholar
- European Medicines Agency (2012) Guideline on real time release testing (formerly guideline on parametric release). https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-real-time-release-testing-formerly-guideline-parametric-release-revision-1_en.pdf. Accessed 03 May 2022
- European Medicines Agency (2014) Guideline on quality of oral modified release products. https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-quality-oral-modified-release-products_en.pdf. Accessed 05 May 2022
- Farooqi S, Yousuf RI, Shoaib MH, Ahmed K, Ansar S et al (2020) Quality by design (QbD)-based numerical and graphical optimization technique for the development of osmotic pump controlled-release metoclopramide HCl tablets. Drug Des Dev Ther 14:5217–5234 ArticleCASGoogle Scholar
- Florence AT (2011) A short history of controlled drug release and an introduction. In: Wilson C, Crowley P (eds) Controlled release in oral drug delivery, 1st edn. Springer, pp 1–26 Google Scholar
- Food and Drug Administration (1997a) Extended release oral dosage forms: development, evaluation, and application of in vitro/in vivo correlations. https://www.fda.gov/media/70939/download. Accessed 03 May 2022
- Food and Drug Administration (1997b) SUPAC-MR: modified release solid oral dosage forms - scale-up and postapproval changes: chemistry, manufacturing, and controls; in vitro dissolution testing and in vivo bioequivalence documentation. https://www.fda.gov/media/70956/download. Accessed 03 May 2022
- Food and Drug Administration (2004) PAT—A framework for innovative pharmaceutical development, manufacturing, and quality assurance. https://www.fda.gov/media/71012/download. Accessed 03 May 2022
- Ford JL (2014) Design and evaluation of hydroxypropyl methylcellulose matrix tablets for oral controlled release: A historical perspective. In: Timmins P, Pygall SR, Melia CD (eds) Hydrophilic matrix tablets for oral controlled release. Springer, pp 17–51 ChapterGoogle Scholar
- Frenning G (2011) Modelling drug release from inert matrix systems: from moving-boundary to continuous-field descriptions. Int J Pharm 418:88–99 ArticleCASPubMedGoogle Scholar
- Galata DL, Farkas A, Konyves Z, Meszaros LA, Szabo E et al (2019) Fast, spectroscopy-based prediction of in vitro dissolution profile of extended release tablets using artificial neural networks. Pharmaceutics 11:400 ArticleCASPubMedPubMed CentralGoogle Scholar
- Galata DL, Konyves Z, Nagy B, Novak M, Meszaros LA et al (2021) Real-time release testing of dissolution based on surrogate models developed by machine learning algorithms using NIR spectra, compression force and particle size distribution as input data. Int J Pharm 597:120338 ArticleCASPubMedGoogle Scholar
- Gavan A, Porfire A, Marina C, Tomuta I (2017) Original research paper. Formulation and pharmaceutical development of quetiapine fumarate sustained release matrix tablets using a QbD approach. Acta Pharm 67:53–70 ArticleCASPubMedGoogle Scholar
- Gavan A, Sylvester B, Porfire A, Iurian S, Casian T et al (2022) NIR spectroscopy for monitoring of the critical manufacturing steps and quality attributes of paliperidone prolonged release tablets. J Mol Struct 1247:131326 ArticleCASGoogle Scholar
- Gendre C, Boiret M, Genty M, Chaminade P, Pean JM (2011) Real-time predictions of drug release and end point detection of a coating operation by in-line near infrared measurements. Int J Pharm 421:237–243 ArticleCASPubMedGoogle Scholar
- Gibson M, Carmody A, Weaver R (2018) Development and manufacture of drug product. In: Schlindwein WS, Gibson M (eds) Pharmaceutical quality by design: a practical approach, 1st edn. John Wiley & Sons, United States, p 117 ChapterGoogle Scholar
- Goodwin DJ, van den Ban S, Denham M, Barylski I (2018) Real time release testing of tablet content and content uniformity. Int J Pharm 537:183–192 ArticleCASPubMedGoogle Scholar
- Gowthami B, Krishna SVG, Rao DS (2021) Formulation of tablets in capsule system: statistical optimization for chronotherapeutic drug delivery of propranolol hydrochloride. J Drug Deliv Sci Technol 63:102398 ArticleCASGoogle Scholar
- Grangeia HB, Silva C, Simoes SP, Reis MS (2020) Quality by design in pharmaceutical manufacturing: a systematic review of current status, challenges and future perspectives. Eur J Pharm Biopharm 147:19–37 ArticleCASPubMedGoogle Scholar
- Guler GK, Eroglu H, Oner L (2017) Development and formulation of floating tablet formulation containing rosiglitazone maleate using Artificial neural network. J Drug Deliv Sci Technol 39:385–397 ArticleCASGoogle Scholar
- Haenlein M, Kaplan A (2019) A brief history of artificial intelligence: on the past, present, and future of artificial intelligence. Calif Manage Rev 61:5–14 ArticleGoogle Scholar
- Hayashi Y, Marumo Y, Takahashi T, Nakano Y, Kosugi A et al (2019) In silico predictions of tablet density using a quantitative structure-property relationship model. Int J Pharm 558:351–356 ArticleCASPubMedGoogle Scholar
- Hayashi Y, Nakano Y, Marumo Y, Kumada S, Okada K et al (2021) Application of machine learning to a material library for modeling of relationships between material properties and tablet properties. Int J Pharm 609:121158 ArticleCASPubMedGoogle Scholar
- Heiman J, Tajarobi F, Gururajan B, Juppo A, Abrahmsen-Alami S (2015) Roller compaction of hydrophilic extended release tablets-combined effects of processing variables and drug/matrix former particle size. AAPS PharmSciTech 16:267–277 ArticleCASPubMedGoogle Scholar
- Heng PWS, Chan LW, Easterbrook MG, Li XM (2001) Investigation of the influence of mean HPMC particle size and number of polymer particles on the release of aspirin from swellable hydrophilic matrix tablets. J Control Release 76:39–49 ArticleCASPubMedGoogle Scholar
- Hiremath PS, Saha RN (2008) Controlled release hydrophilic matrix tablet formulations of isoniazid: design and in vitro studies. AAPS PharmSciTech 9:1171–1178 ArticleCASPubMedPubMed CentralGoogle Scholar
- Hoffman AS (2008) The origins and evolution of “controlled” drug delivery systems. J Control Release 132:153–163 ArticleCASPubMedGoogle Scholar
- Hu M, Zhu Z, Wu Y, Meng Q, Luo J et al (2020) Exploring the potential of hydrophilic matrix combined with insoluble film coating: preparation and evaluation of ambroxol hydrochloride extended release tablets. AAPS PharmSciTech 21:93 ArticleCASPubMedGoogle Scholar
- Huang J, Kaul G, Cai CS, Chatlapalli R, Hernandez-Abad P et al (2009) Quality by design case study: an integrated multivariate approach to drug product and process development. Int J Pharm 382:23–32 ArticleCASPubMedGoogle Scholar
- Ibric S, Jovanovic M, Djuric Z, Parojcic J, Solomun L et al (2007) Generalized regression neural networks in prediction of drug stability. J Pharm Pharmacol 59:745–750 ArticleCASPubMedGoogle Scholar
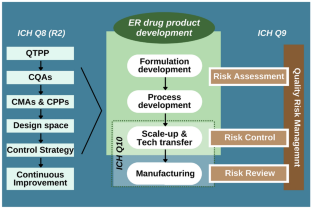
- International Conference on Harmonisation (2009) ICH guideline Q8 (R2) on pharmaceutical development. https://database.ich.org/sites/default/files/Q8_R2_Guideline.pdf. Accessed 03 May 2022
- International Conference on Harmonisation (2010) Q8, Q9 and Q10: Questions and answers. https://database.ich.org/sites/default/files/Q8_Q9_Q10_Q%26As_R4_Q%26As_0.pdf. Accessed 06 May 2022
- International Conference on Harmonisation (2011) Q8, Q9 and Q10: Points to consider. https://database.ich.org/sites/default/files/Q8_Q9_Q10_Q%26As_R4_Points_to_Consider_2.pdf. Accessed 06 May 2022
- International Conference on Harmonisation (2012) ICH guideline Q11 on development and manufacture of drug substances (chemical entities and biotechnological/biological entities). https://database.ich.org/sites/default/files/Q11%20Guideline.pdf. Accessed 03 May 2022
- Ilic M, Duris J, Kovacevic I, Ibric S, Parojcic J (2014) In vitro—in silico—in vivo drug absorption model development based on mechanistic gastrointestinal simulation and artificial neural networks: Nifedipine osmotic release tablets case study. Eur J Pharm Sci 62:212–218 ArticleCASPubMedGoogle Scholar
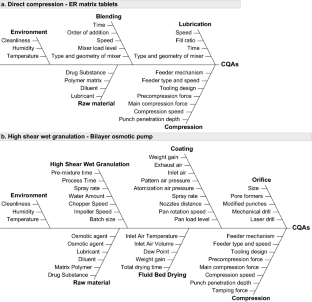
- Ilyes K, Casian T, Hales D, Borodi G, Rus L et al (2021) Applying the principles of quality by design (Qbd) coupled with multivariate data analysis (MVDA) in establishing the impact of raw material variability for extended release tablets. Farmacia 69:481–497 ArticleCASGoogle Scholar
- International Conference on Harmonisation (2005) ICH guideline Q9 on quality risk management. https://database.ich.org/sites/default/files/Q9%20Guideline.pdf. Accessed 03 May 2022
- International Conference on Harmonisation (2008) ICH guideline Q10 on pharmaceutical quality system. https://database.ich.org/sites/default/files/Q10%20Guideline.pdf. Accessed 03 May 2022
- International Conference on Harmonisation (2019) ICH guideline Q12 on technical and regulatory considerations for pharmaceutical product lifecycle management. https://database.ich.org/sites/default/files/Q12_Guideline_Step4_2019_1119.pdf. Accessed 03 May 2022
- Islam MT, Maniruzzaman M, Halsey SA, Chowdhry BZ, Douroumis D (2014) Development of sustained-release formulations processed by hot-melt extrusion by using a quality-by-design approach. Drug Deliv Transl Res 4:377–387 ArticleCASPubMedGoogle Scholar
- Iurian S, Turdean L, Tomuta I (2017) Risk assessment and experimental design in the development of a prolonged release drug delivery system with paliperidone. Drug Des Dev Ther 11:733–746 ArticleCASGoogle Scholar
- Ivic B, Ibric S, Betz G, Djuric Z (2010) Optimization of drug release from compressed multi unit particle system (MUPS) using generalized regression neural network (GRNN). Arch Pharm Res 33:103–113 ArticleCASPubMedGoogle Scholar
- Jang EH, Park YS, Choi D (2021) Investigation of the effects of materials and dry granulation process on the mirabegron tablet by integrated QbD approach with multivariate analysis. Powder Technol 382:23–39 ArticleCASGoogle Scholar
- Kanwal U, Mukhtar S, Waheed M, Mehreen A, Abbas N et al (2021) Fixed dose single tablet formulation with differential release of amlodipine besylate and simvastatin and its pharmacokinetic profile: QbD and risk assessment approach. Drug Des Dev Ther 15:2193–2210 ArticleGoogle Scholar
- Khan AM, Hanif M, Bukhari NI, Shamim R, Rasool F et al (2020) Artificial neural network (ANN) approach to predict an optimized pH-dependent mesalamine matrix tablet. Drug Des Dev Ther 14:2435–2448 ArticleCASGoogle Scholar
- Kim CJ (1998) Effects of drug solubility, drug loading, and polymer molecular weight on drug release from polyox (R) tablets. Drug Dev Ind Pharm 24:645–651 ArticleCASPubMedGoogle Scholar
- Kosir D, Ojstersek T, Baumgartner S, Vrecer F (2018) A study of critical functionality-related characteristics of HPMC for sustained-release tablets. Pharm Dev Technol 23:865–873 ArticleCASPubMedGoogle Scholar
- Kovacs B, Peterfi O, Kovacs-Deak B, Szekely-Szentmiklosi I, Fulop I et al (2021) Quality-by-design in pharmaceutical development: from current perspectives to practical applications. Acta Pharm 71:497–526 ArticleCASPubMedGoogle Scholar
- Kushner J, Lamba M, Stock T, Wang R, Nemeth MA et al (2020) Development and validation of a Level A in vitro in vivo correlation for tofacitinib modified -release tablets using extrudable core system osmotic delivery technology. Eur J Pharm Sci 147:105200 ArticleCASPubMedGoogle Scholar
- Lakio S, Tajarobi P, Wikstrom H, Fransson M, Arnehed J et al (2016) Achieving a robust drug release from extended release tablets using an integrated continuous mixing and direct compression line. Int J Pharm 511:659–668 ArticleCASPubMedGoogle Scholar
- Lee PI, Li JX (2010) Evolution of oral controlled release dosage forms. In: Wen H, Park K (eds) Oral controlled release formulation design and drug delivery: theory to practice, 1st edn. John Wiley & Sons, New Jersey, pp 21–31 ChapterGoogle Scholar
- Lefnaoui S, Rebouh S, Bouhedda M, Yahoum MM, Hanini S (2018) Artificial neural network modeling of sustained antihypertensive drug delivery using polyelectrolyte complex based on carboxymethyl-kappa-carrageenan and chitosan as prospective carriers. International conference on applied smart systems (ICASS), USA BookGoogle Scholar
- Li H, Hardy RJ, Gu X (2008) Effect of drug solubility on polymer hydration and drug dissolution from polyethylene oxide (PEO) matrix tablets. AAPS PharmSciTech 9:437–443 ArticleCASPubMedPubMed CentralGoogle Scholar
- Lin W, Li YK, Shi QZ, Liao XR, Zeng Y et al (2022) Preparation and evaluation of bilayer-core osmotic pump tablets contained topiramate. PLoS ONE 17:e0264457 ArticleCASPubMedPubMed CentralGoogle Scholar
- Liu LX, Xu XN (2008) Preparation of bilayer-core osmotic pump tablet by coating the indented core tablet. Int J Pharm 352:225–230 ArticleCASPubMedGoogle Scholar
- Liu LL, Zhang KF, Sun ZY, Dong Q, Li L et al (2021) A new perspective in understanding the dissolution behavior of nifedipine controlled release tablets by NIR spectroscopy with aquaphotomics. J Mol Struct 1230:129872 ArticleCASGoogle Scholar
- Lopes JA, Sarraguca MC (2018) Data processing in multivariate analysis of pharmaceutical processes. In: Ferreira A, Menezes J, Tobyn M (eds) Multivariate analysis in the pharmaceutical industry, 1st edn. Elsevier, United Kingdom, pp 35–51 Google Scholar
- Lou H, Chung JI, Kiang YH, Xiao LY, Hageman MJ (2019) The application of machine learning algorithms in understanding the effect of core/shell technique on improving powder compactability. Int J Pharm 555:368–379 ArticleCASPubMedGoogle Scholar
- Lou H, Lian B, Hageman MJ (2021) Applications of machine learning in solid oral dosage form development. J Pharm Sci 110:3150–3165 ArticleCASPubMedGoogle Scholar
- Lundsberg-Nielsen L, Schlindwein WS, Berghaus A (2018) Process analytical technology (PAT). In: Schlindwein WS, Gibson M (eds) Pharmaceutical quality by design: a practical approach, 1st edn. John Wiley & Sons, United States, pp 227–255 ChapterGoogle Scholar
- Maderuelo C, Zarzuelo A, Lanao JM (2011) Critical factors in the release of drugs from sustained release hydrophilic matrices. J Control Release 154:2–19 ArticleCASPubMedGoogle Scholar
- Maki-Lohiluoma E, Sakkinen N, Palomaki M, Winberg O, Ta HX et al (2021) Use of machine learning in prediction of granule particle size distribution and tablet tensile strength in commercial pharmaceutical manufacturing. Int J Pharm 609:121146 ArticleCASPubMedGoogle Scholar
- Malaterre V, Ogorka J, Loggia N, Gurny R (2009) Approach to design push-pull osmotic pumps. Int J Pharm 376:56–62 ArticleCASPubMedGoogle Scholar
- Markl D, Warman M, Dumarey M, Bergman EL, Folestad S et al (2020) Review of real-time release testing of pharmaceutical tablets: state-of-the art, challenges and future perspective. Int J Pharm 582:119353 ArticleCASPubMedGoogle Scholar
- Mirani AG, Patankar SP, Kadam VJ (2016) Risk-based approach for systematic development of gastroretentive drug delivery system. Drug Deliv Transl Res 6:579–596 ArticleCASPubMedGoogle Scholar
- Missaghi S, Patel P, Farrell TP, Huatan H, Rajabi-Siahboomi AR (2014) Investigation of critical core formulation and process parameters for osmotic pump oral drug delivery. AAPS PharmSciTech 15:149–160 ArticleCASPubMedGoogle Scholar
- Mohamed MI, Al-Mahallawi AM, Awadalla SM (2020) Development and optimization of osmotically controlled drug delivery system for poorly aqueous soluble diacerein to improve its bioavailability. Drug Dev Ind Pharm 46:814–825 ArticleCASPubMedGoogle Scholar
- Momin MM, Kane S, Abhang P (2015) Formulation and evaluation of bilayer tablet for bimodal release of venlafaxine hydrochloride. Front Pharmacol 6:144 ArticlePubMedPubMed CentralGoogle Scholar
- Muller J, Brock D, Knop K, Zeitler JA, Kleinebudde P (2012) Prediction of dissolution time and coating thickness of sustained release formulations using Raman spectroscopy and terahertz pulsed imaging. Eur J Pharm Biopharm 80:690–697 ArticlePubMedGoogle Scholar
- Muntean DM, Alecu C, Tomuta I (2017) Simultaneous quantification of paracetamol and caffeine in powder blends for tableting by NIR-chemometry. J Spectrosc 2017:7160675 ArticleGoogle Scholar
- Nagy B, Petra D, Galata DL, Demuth B, Borbas E et al (2019) Application of artificial neural networks for process analytical technology-based dissolution testing. Int J Pharm 567:118464 ArticleCASPubMedGoogle Scholar
- Nokhodchi A, Raja S, Patel P, Asare-Addo K (2012) The role of oral controlled release matrix tablets in drug delivery systems. Bioimpacts 2:175–187 CASPubMedPubMed CentralGoogle Scholar
- Obeidat WM, Nokhodchi A, Alkhatib H (2015) Evaluation of matrix tablets based on Eudragit®E100/Carbopol®971P combinations for controlled release and improved compaction properties of water soluble model drug paracetamol. AAPS PharmSciTech 16:1169–1179 ArticleCASPubMedPubMed CentralGoogle Scholar
- Owen M, Cox I (2018) Design of experiments. In: Schlindwein WS, Gibson M (eds) Pharmaceutical quality by design: a practical approach, 1st edn. John Wiley & Sons, United States, pp 157–199 ChapterGoogle Scholar
- Parmar C, Parikh K, Mundada P, Bhavsar D, Sawant K (2018) Formulation and optimization of enteric coated bilayer tablets of mesalamine by RSM: In vitro—In vivo investigations and roentogenographic study. J Drug Deliv Sci Technol 44:388–398 ArticleCASGoogle Scholar
- Paul D, Sanap G, Shenoy S, Kalyane D, Kalia K et al (2021a) Artificial intelligence in drug discovery and development. Drug Discov Today 26:80–93 ArticleCASPubMedGoogle Scholar
- Paul S, Baranwal Y, Tseng YC (2021b) An insight into predictive parameters of tablet capping by machine learning and multivariate tools. Int J Pharm 599:120439 ArticleCASPubMedGoogle Scholar
- Pawar P, Wang YF, Keyvan G, Callegari G, Cuitino A et al (2016) Enabling real time release testing by NIR prediction of dissolution of tablets made by continuous direct compression (CDC). Int J Pharm 512:96–107 ArticleCASPubMedGoogle Scholar
- Peppas NA, Narasimhan B (2014) Mathematical models in drug delivery: how modeling has shaped the way we design new drug delivery systems. J Control Release 190:75–81 ArticleCASPubMedGoogle Scholar
- Petrovic J, Ibric S, Betz G, Duric Z (2012) Optimization of matrix tablets controlled drug release using Elman dynamic neural networks and decision trees. Int J Pharm 428:57–67 ArticleCASPubMedGoogle Scholar
- Pishnamazi M, Ismail HY, Shirazian S, Iqbal J, Walker GM et al (2019) Application of lignin in controlled release: development of predictive model based on artificial neural network for API release. Cellulose 26:6165–6178 ArticleCASGoogle Scholar
- Politis SN, Colombo P, Colombo G (2017) Design of experiments (DoE) in pharmaceutical development. Drug Dev Ind Pharm 43:889–901 ArticleCASGoogle Scholar
- Porfire A, Filip C, Tomuta I (2017) High-throughput NIR-chemometric methods for chemical and pharmaceutical characterization of sustained release tablets. J Pharm Biomed Anal 138:1–13 ArticleCASPubMedGoogle Scholar
- Qazi F, Shoaib MH, Yousuf RI, Siddiqui F, Nasiri MI et al (2020) QbD based Eudragit coated meclizine HCl immediate and extended release multiparticulates: formulation, characterization and pharmacokinetic evaluation using HPLC-Fluorescence detection method. Sci Rep 10:14765 ArticleCASPubMedPubMed CentralGoogle Scholar
- Qiu Y, Lee P (2017) Rational design of oral modified-release drug delivery systems. In: Qiu Y, Chen Y, Zhang G, Yu L, Mantri R (eds) Developing solid oral dosage forms, 2nd edn. Academic Press, London, pp 519–554 ChapterGoogle Scholar
- Rajalahti T, Kvalheim OM (2011) Multivariate data analysis in pharmaceutics: a tutorial review. Int J Pharm 417:280–290 ArticleCASPubMedGoogle Scholar
- Reda C, Kaufmann E, Delahaye-Duriez A (2020) Machine learning applications in drug development. Comput Struct Biotechnol J 18:241–252 ArticleCASPubMedGoogle Scholar
- Reinhardt IC, Oliveira JC, Ring DT (2020) Current perspectives on the development of industry 4.0 in the pharmaceutical sector. J Ind Inf Integr 18:100131 Google Scholar
- Reynolds TD, Mitchell SA, Balwinski KM (2002) Investigation of the effect of tablet surface area/volume on drug release from hydroxypropylmethylcellulose controlled-release matrix tablets. Drug Dev Ind Pharm 28:457–466 ArticleCASPubMedGoogle Scholar
- Rus LL, Casian T, Iovanov RI, Orzea RM, Onisor I et al (2020) Quantitative characterization of sustained release tablets with diclofenac sodium by means of near-infrares spectroscopy and chemometry. Farmacia 68:728–739 ArticleCASGoogle Scholar
- Sacher S, Poms J, Rehrl J, Khinast JG (2022) PAT implementation for advanced process control in solid dosage manufacturing-A practical guide. Int J Pharm 613:121408 ArticleCASPubMedGoogle Scholar
- Sanoufi MR, Aljaberi A, Hamdan I, Al-Zoubi N (2020) The use of design of experiments to develop hot melt extrudates for extended release of diclofenac sodium. Pharm Dev Technol 25:187–196 ArticleCASPubMedGoogle Scholar
- Saracoglu OK, Uludag MO, Ozdemir ED, Degim IT (2020) Development of controlled release dexketoprofen tablets and prediction of drug release using artificial neural network (ANN) modelling. Braz J Pharm Sci 56:e18540 ArticleCASGoogle Scholar
- Saydam M, Takka S (2018) Development and in vitro evaluation of pH-independent release matrix tablet of weakly acidic drug valsartan using quality by design tools. Drug Dev Ind Pharm 44:1905–1917 ArticleCASPubMedGoogle Scholar
- Schmitt S (2018) Quality systems and knowledge management. In: Schlindwein WS, Gibson M (eds) Pharmaceutical quality by design: a practical approach, 1st edn. John Wiley & Sons, United States, pp 47–60 ChapterGoogle Scholar
- Sethi S, Mangla B, Kamboj S, Rana V (2018) A QbD approach for the fabrication of immediate and prolong buoyant cinnarizine tablet using polyacrylamide-g-corn fibre gum. Int J Biol Macromol 117:350–361 ArticleCASPubMedGoogle Scholar
- Shah V, Khambhla E, Nivsarkar M, Trivedi R, Patel RK (2022) An Integrative QbD approach for the development and optimization of controlled release compressed coated formulation of water-soluble drugs. AAPS PharmSciTech 23:120 ArticleCASPubMedGoogle Scholar
- Shi GL, Lin LF, Liu YL, Chen GS, Luo YT et al (2021) Pharmaceutical application of multivariate modelling techniques: a review on the manufacturing of tablets. RSC Adv 11:8323–8345 ArticleCASPubMedPubMed CentralGoogle Scholar
- Siegel RA, Rathbone MJ (2012) Overview of controlled release mechanisms. In: Siepmann J, Siegel RA, Rathbone MJ (eds) Fundamentals and applications of controlled release drug delivery, 1st edn. Springer, New York, pp 19–43 ChapterGoogle Scholar
- Siepmann J, Siepmann F (2012b) Swelling controlled drug delivery systems. In: Rathbone MJ, Siepmann J, Siegel RA (eds) Fundamentals and applications of controlled release drug delivery, 1st edn. Springer, New York, pp 153–170 ChapterGoogle Scholar
- Siepmann J, Kranz H, Peppas NA, Bodmeier R (2000) Calculation of the required size and shape of hydroxypropyl methylcellulose matrices to achieve desired drug release profiles. Int J Pharm 201:151–164 ArticleCASPubMedGoogle Scholar
- Siepmann J, Siegel RA, Siepmann F (2012a) Diffusion controlled drug delivery systems. In: Rathbone MJ, Siepmann J, Siegel RA (eds) Fundamentals and applications of controlled release drug delivery, 1st edn. Springer, New York, pp 127–152 ChapterGoogle Scholar
- Simoes MF, Silva G, Pinto AC, Fonseca M, Silva NE et al (2020) Artificial neural networks applied to quality-by-design: From formulation development to clinical outcome. Eur J Pharm Biopharm 152:282–295 ArticleCASPubMedGoogle Scholar
- Singh SK, Venkateshwarn T, Simmons SP (2010) Oral controlled drug delivery: quality by design (QbD) approach to drug development. In: Wen H, Park K (eds) Oral controlled release formulation design and drug delivery: theory to practice, 1st edn. John Wiley & Sons, New Jersey, pp 279–303 ChapterGoogle Scholar
- Sirbu C, Tomuta I, Achim M, Rus LL, Vonica L et al (2014) Quantitative characterization of powder blends for tablets with Indapamide by near-infrared spectrocopy and chemometry. Farmacia 62:48–57 CASGoogle Scholar
- Steinwandter V, Borchert D, Herwig C (2019) Data science tools and applications on the way to Pharma 4.0. Drug Discov Today 24:1795–1805 ArticlePubMedGoogle Scholar
- Than YM, Titapiwatanakun V (2021) Statistical design of experiment-based formulation development and optimization of 3D printed oral controlled release drug delivery with multi target product profile. J Pharm Investig 51:715–734 ArticleCASGoogle Scholar
- Thapa P, Jeong SH (2018) Effects of formulation and process variables on gastroretentive floating tablets with a high-dose soluble drug and experimental design approach. Pharmaceutics 10:161 ArticleCASPubMedPubMed CentralGoogle Scholar
- Theeuwes F (1975) Elementary osmotic pumps. J Pharm Sci 64:1987–1991 ArticleCASPubMedGoogle Scholar
- Thomas S, Palahnuk H, Amini H, Akseli I (2021) Data-smart machine learning methods for predicting composition-dependent Young’s modulus of pharmaceutical compacts. Int J Pharm 592:120049 ArticleCASPubMedGoogle Scholar
- Timmins P, Desai D, Chen W, Wray P, Brown J et al (2016) Advances in mechanistic understanding of release rate control mechanisms of extended-release hydrophilic matrix tablets. Ther Deliv 7:553–572 ArticleCASPubMedGoogle Scholar
- Tiwari SB, DiNunzio J, Rajabi-Siahboomi A (2011) Drug–polymer matrices for extended release. In: Wilson CG, Crowley PJ (eds) Controlled release in oral drug delivery, 1st edn. Springer, New York, pp 131–159 ChapterGoogle Scholar
- Vamathevan J, Clark D, Czodrowski P, Dunham I, Ferran E et al (2019) Applications of machine learning in drug discovery and development. Nat Rev Drug Discov 18:463–477 ArticleCASPubMedPubMed CentralGoogle Scholar
- Van Hauwermeiren D, Stock M, De Beer T, Nopens I (2020) Predicting pharmaceutical particle size distributions using kernel mean embedding. Pharmaceutics 12:271 ArticlePubMedPubMed CentralGoogle Scholar
- Van Snick B, Holman J, Cunningham C, Kumar A, Vercruysse J et al (2017) Continuous direct compression as manufacturing platform for sustained release tablets. Int J Pharm 519:390–407 ArticlePubMedGoogle Scholar
- Vanhoorne V, Vanbillemont B, Vercruysse J, De Leersnyder F, Gomes P et al (2016) Development of a controlled release formulation by continuous twin screw granulation: Influence of process and formulation parameters. Int J Pharm 505:61–68 ArticleCASPubMedGoogle Scholar
- Vanza JD, Patel RB, Dave RR, Patel MR (2020) Polyethylene oxide and its controlled release properties in hydrophilic matrix tablets for oral administration. Pharm Dev Technol 25:1–19 ArticleGoogle Scholar
- Verma RK, Krishna DM, Garg S (2002) Formulation aspects in the development of osmotically controlled oral drug delivery systems. J Control Release 79:7–27 ArticleCASPubMedGoogle Scholar
- Viriden A, Wittgren B, Andersson T, Larsson A (2009) The effect of chemical heterogeneity of HPMC on polymer release from matrix tablets. Eur J Pharm Sci 36:392–400 ArticleCASPubMedGoogle Scholar
- Viriden A, Larsson A, Wittgren B (2010) The effect of substitution pattern of HPMC on polymer release from matrix tablets. Int J Pharm 389:147–156 ArticleCASPubMedGoogle Scholar
- Viriden A, Abrahmsen-Alami S, Wittgren B, Larsson A (2011) Release of theophylline and carbamazepine from matrix tablets–consequences of HPMC chemical heterogeneity. Eur J Pharm Biopharm 78:470–479 ArticleCASPubMedGoogle Scholar
- Vora C, Patadia R, Mittal K, Mashru R (2015) Risk based approach for design and optimization of site specific delivery of isoniazid. J Pharm Investig 45:249–264 ArticleCASGoogle Scholar
- Walker RB (2008) Modified-release delivery systems for oral use. In: Rathbone M, Hadgraft J, Roberts MS, Lane ME (eds) Modified-release drug delivery technology, 2nd edn. CRC Press, New York, pp 131–141 Google Scholar
- Wang W, Ye ZYF, Gao HL, Ouyang DF (2021) Computational pharmaceutics-A new paradigm of drug delivery. J Control Release 338:119–136 ArticleCASPubMedGoogle Scholar
- Wang S, Di JW, Wang D, Dai XD, Hua YB et al (2022) State-of-the-art review of artificial neural networks to predict. Characterize and optimize pharmaceutical formulation. Pharmaceutics 14:183 ArticlePubMedPubMed CentralGoogle Scholar
- Wen H, Park K (2010) Introduction and overview of oral controlled release formulation design. In: Wen H, Park K (eds) Oral Controlled release formulation design and drug delivery: theory to practice, 1st edn. John Wiley & Sons, New Jersey, pp 1–19 ChapterGoogle Scholar
- Wirges M, Funke A, Serno P, Knop K, Kleinebudde P (2013) Monitoring of an active coating process for two-layer tablets-model development strategies. J Pharm Sci 102:556–564 ArticleCASPubMedGoogle Scholar
- Won DH, Park H, Ha ES, Kim HH, Jang SW et al (2021) Optimization of bilayer tablet manufacturing process for fixed dose combination of sustained release high-dose drug and immediate release low-dose drug based on quality by design (QbD). Int J Pharm 605:120838 ArticleCASPubMedGoogle Scholar
- Wu HQ, Lyon RC, Khan MA, Voytilla RJ, Drennen JK (2015) Integration of near-infrared spectroscopy and mechanistic modeling for predicting film-coating and dissolution of modified release tablets. Ind Eng Chem Res 54:6012–6023 ArticleCASGoogle Scholar
- Yang Y, Zhao ZN, Wang YF, Yang L, Liu DD et al (2016) A novel asymmetric membrane osmotic pump capsule with in situ formed delivery orifices for controlled release of gliclazide solid dispersion system. Int J Pharm 506:340–350 ArticleCASPubMedGoogle Scholar
- Yang YL, Ye ZYF, Su Y, Zhao QQ, Li XS et al (2019) Deep learning for in vitro prediction of pharmaceutical formulations. Acta Pharm Sin B 9:177–185 ArticlePubMedGoogle Scholar
- Yoo S, Kim J, Choi GJ (2022) Drug properties prediction based on deep learning. Pharmaceutics 14:467 ArticlePubMedPubMed CentralGoogle Scholar
- Yu LX (2008) Pharmaceutical quality by design: product and process development, understanding, and control. Pharm Res 25:781–791 ArticleCASPubMedGoogle Scholar
- Yu LX, Amidon G, Khan MA, Hoag SW, Polli J et al (2014) Understanding pharmaceutical quality by design. AAPS J 16:771–783 ArticleCASPubMedPubMed CentralGoogle Scholar
- Yu JJ, Meng X, Dong X, Han MF, Li GT et al (2021) Synthesis and characterization of osmotic pump capsules containing polyoxyethylene and pH modifier to control the release of nifedipine. Eur J Pharm Biopharm 163:102–108 ArticleCASPubMedGoogle Scholar
- Zaborenko N, Shi Z, Corredor CC, Smith-Goettler BM, Zhang L et al (2019) First-principles and empirical approaches to predicting in vitro dissolution for pharmaceutical formulation and process development and for product release testing. AAPS J 21:32 ArticlePubMedGoogle Scholar
- Zarmpi P, Flanagan T, Meehan E, Mann J, Fotaki N (2017) Biopharmaceutical aspects and implications of excipient variability in drug product performance. Eur J Pharm Biopharm 111:1–15 ArticleCASPubMedGoogle Scholar
- Zhou DL, Law D, Reynolds J, Davis L, Smith C et al (2014) Understanding and managing the impact of HPMC variability on drug release from controlled release formulations. J Pharm Sci 103:1664–1672 ArticleCASPubMedGoogle Scholar
Acknowledgements
This work was financially supported by the Drugs R&D Doctoral Program assigned by FCT (Fundação para a Ciência e Tecnologia), Portugal and Tecnimede Group [grant PD/BDE/150736/2020].








